













Product Introduction
Contaminant concerns have become a decisive factor in the global honey trade. Importing countries increasingly enforce strict residue limits for heavy metals (lead, cadmium, mercury), veterinary antibiotics (chloramphenicol, tetracyclines, sulfonamides, nitrofurans, quinolones), and pesticide residues — and honey shipments that fail to meet these thresholds face rejection, destruction, or costly returns. At the same time, conventional quality parameters such as HMF, diastase number, and moisture remain essential for verifying that honey has not been adulterated or degraded during processing and storage.
For honey producers, traders, and regulatory authorities, the practical challenge is that quantitative quality testing and qualitative contaminant screening typically require different analytical methods and separate instruments — colorimetric analysis for quality parameters and immunochromatographic assays for residue detection. This fragmentation increases equipment costs, extends testing timelines, and complicates data management across the quality control workflow.
The HM-FM2 resolves this fragmentation by combining photoelectric colorimetric and lateral flow immunochromatographic detection modules in a single portable instrument. The colorimetric module quantifies 9 quality parameters — sucrose, reducing sugars, HMF, diastase number, proline, acidity, moisture, sugar content, and ash — through 12 simultaneous detection channels with an absorbance range of 0–3.5 OD. The immunochromatographic module performs qualitative screening for pesticide residues, heavy metals, and antibiotics using colloidal gold test cards, with automatic scanning, C/T line recognition, and card ejection, and is compatible with commercially available non-original cards.
The dual-module design enables laboratories and inspection stations to complete both quality assessment and contaminant screening in a single workflow with one instrument, reducing capital expenditure and operational complexity. AC/DC dual power supply with vehicle adapter capability supports deployment at purchasing stations, border crossings, and mobile inspection units. Wi-Fi cloud-based remote software upgrade ensures that detection methods stay current as regulatory limits evolve, and USB data export to Excel facilitates integration with existing quality management systems.
Applications
Honey purchasing and collection stations: on-site quality and contaminant screening before acceptance
Honey processing facilities: incoming raw material inspection covering both quality and safety parameters
Import/export inspection and quarantine: comprehensive residue screening for trade clearance
Market supervision and regulation: enforcement testing against national honey safety standards
Quality supervision and testing institutions: laboratory-grade analysis for certification and audit
Mobile inspection units: portable field deployment at border checkpoints and trade fairs
Key Features & Advantages
One-instrument dual detection: photoelectric colorimetry + lateral flow immunochromatography eliminates the need for separate devices for quality and residue testing
Qualitative screening for heavy metals (lead, cadmium, mercury) via immunochromatographic strips
Qualitative screening for antibiotics (chloramphenicol, tetracyclines, sulfonamides, nitrofurans, quinolones) using colloidal gold test cards
Automatic colloidal gold card scanning, C/T line recognition, and automatic card ejection for streamlined immunochromatographic workflow
Compatibility with commercially available non-original colloidal gold cards for procurement flexibility
9 quantitative quality parameters measured through 12 simultaneous channels with 0–3.5 OD absorbance range
Four-wavelength cold light sources per channel with automatic brightness adjustment for stable colorimetric performance
Colorimetric result threshold lines modifiable with reference values retained after power loss for customized quality criteria
AC/DC dual power with built-in lithium battery and vehicle power adapter for mobile inspection deployment
Wi-Fi cloud-based remote software upgrade for keeping pace with evolving regulatory residue limits
Built-in thermal printer and USB export to Excel for immediate documentation and structured data management
Android 7-inch color touchscreen with integrated dual-module operation for straightforward workflow management
Automatic self-check, zero calibration, and repeatability detection for measurement confidence
Multi-category sample menu library with customizable database entries for diverse honey types and origins
Single-well repeatability ≤0.01 OD and channel variation ≤0.08 OD for precise colorimetric results
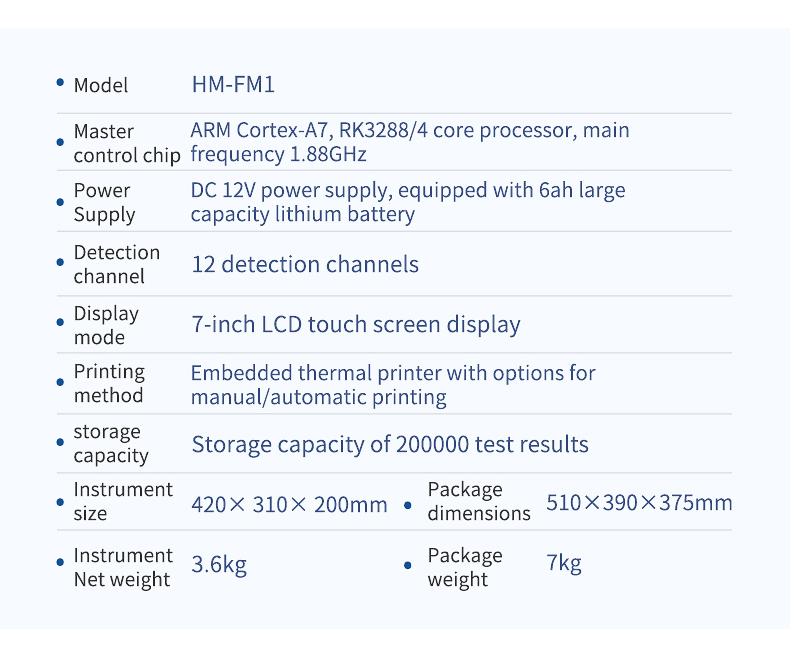
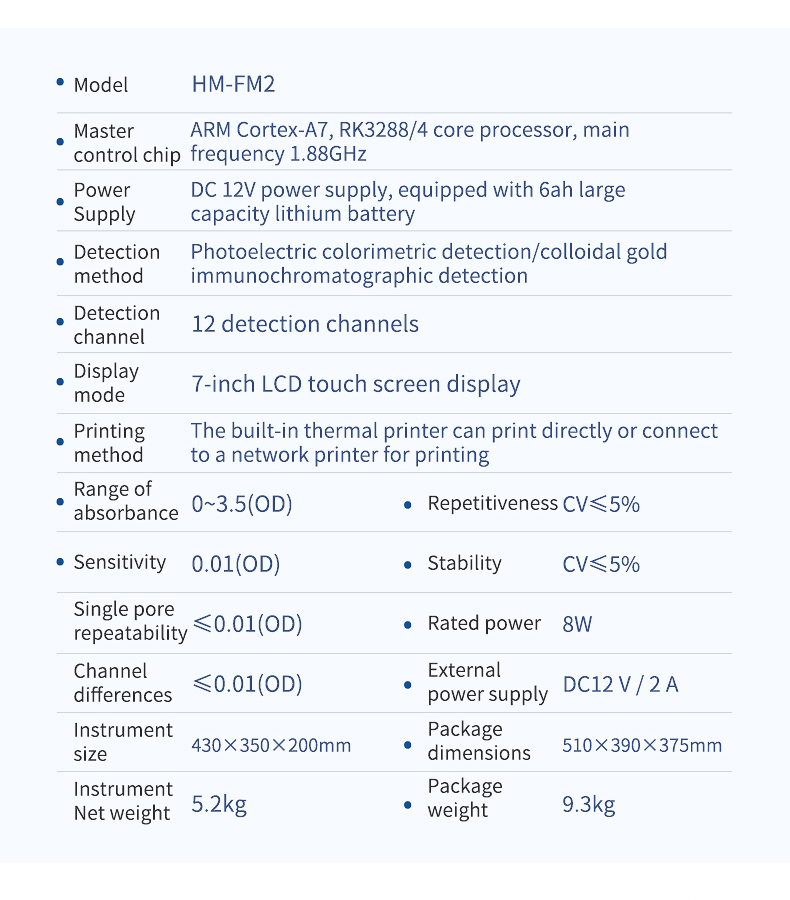
Technical Specifications
| Parameter | Specification |
|---|---|
| Model | HM-FM2 |
| Absorbance Range | 0–3.5 (OD) |
| Sensitivity | 0.01 (OD) |
| Single-Well Repeatability | ≤0.01 (OD) |
| Channel Variation | ≤0.08 (OD) |
| Repeatability | CV ≤5% |
| Stability | CV ≤5% |
| Detection Channels | 12 |
| Display | 7-inch color touchscreen |
| Rated Power | 8W |
| External Power Supply | DC 12V/2A |
| Instrument Dimensions | 430 × 350 × 200 mm |
| Instrument Net Weight | 5.2 kg |
FAQ
Q1: What types of honey contaminants can the HM-FM2 detect?
The HM-FM2 performs qualitative screening for three major contaminant categories through its lateral flow immunochromatographic module: pesticide residues, heavy metals (lead, cadmium, mercury), and antibiotics (chloramphenicol, tetracyclines, sulfonamides, nitrofurans, quinolones). These are the most commonly regulated residue categories in international honey trade. The colorimetric module simultaneously provides quantitative measurements of 9 quality parameters including sucrose, HMF, diastase, and moisture for comprehensive safety assessment.
Q2: How does the lateral flow immunochromatographic module work on the HM-FM2?
The operator inserts a colloidal gold test card into the immunochromatographic module after applying the prepared sample. The module automatically scans the card, identifies the control (C) line and test (T) line, and ejects the card upon completion. C/T line recognition determines the presence or absence of the target analyte. The module is compatible with commercially available non-original colloidal gold cards, giving laboratories flexibility in selecting test card suppliers without being restricted to proprietary consumables.
Q3: Can the HM-FM2 be used for import/export honey inspection?
Yes. The HM-FM2 covers both the quality parameters and residue screening categories typically required by importing country regulations. Its dual-module design allows inspectors to complete quality assessment and contaminant screening in a single workflow, reducing the time needed for clearance decisions. The built-in thermal printer provides immediate documentation, and USB export to Excel facilitates preparation of inspection reports for customs and quarantine authorities.
Q4: What is the advantage of combining colorimetric and immunochromatographic detection in one instrument?
Traditionally, honey quality testing and contaminant screening require separate instruments using fundamentally different detection principles — photoelectric colorimetry for quality parameters and lateral flow immunoassay for residue detection. The HM-FM2's dual-module integration eliminates the need to purchase, maintain, and calibrate two separate devices, reduces the laboratory footprint, and simplifies the testing workflow. Results from both modules are managed through a unified Android interface.
Q5: How does the HM-FM2 support field deployment at purchasing stations?
The HM-FM2 supports AC/DC dual power supply with a built-in lithium battery and a vehicle power adapter, enabling operation at purchasing stations, border checkpoints, and mobile inspection units where grid power may be unreliable or unavailable. The 7-inch touchscreen interface requires minimal operator training, and the built-in thermal printer produces on-site documentation for immediate decision-making during honey purchasing transactions.
Q6: What does Wi-Fi cloud-based remote software upgrade mean for the HM-FM2?
Regulatory limits for honey residues are periodically updated as new scientific evidence emerges. The HM-FM2's Wi-Fi cloud upgrade capability allows the instrument's detection methods and threshold databases to be updated remotely without requiring a technician visit or manual software installation. This ensures that the instrument remains current with the latest residue limits and detection protocols, which is critical for laboratories performing compliance testing against evolving standards.
Article address:https://www.hmfoodtesting.com/sjyp3/honey-safety-screening.html

 Current
location:
Current
location:
 +86
17853698681
+86
17853698681









